:max_bytes(150000):strip_icc()/PeriodicTable-56a12c983df78cf772682271.png)
One copper, one sulfur, 10 hydrogen, nine oxygen. (e) Al 2(SO 4) 3 Two aluminum, three sulfur, and 12 oxygen. Note how nitrogen is in two different places in the formula. (d) NH 4NO 3 Two nitrogen, four H and three O. (c) Al(NO 3) 3 One Al, three nitrogen, and nine oxygens. (b) Fe 2O 3 There are two irons and three oxygens. If parentheses are involved, you must multiply each subscript inside by the one which is outside.Įxample #1: How many of each element are in the following examples? If a subscript follows an atom with no parenthesis, that number tells you how many of that atom are present. 
Mg(OH) 2 has one atom of magnesiun and two each of oxygen and hydrogen. H 2O 2 has two atoms each of oxygen and hydrogen. Point #1: You need to know how many atoms of each element are in a substance in order to calculate its molecular weight.įor example H 2O has two atoms of hydrogen and one atom of oxygen. There are many Internet sites that allow you to input a formula and get the molecular weight. You should have a periodic table for looking up atomic weights and a calculator. The mole is the standard method in chemistry for communicating how much of a substance is present. The molecular weight of a substance is needed to tell us how many grams are in one mole of that substance. If you weigh twice as much, you weigh 24. For example, if you weigh 1/2 as much as C-12, you weigh 6. This is the starting point for how much an atom weighs. (And, of course, amu still exists in many older documents pre-dating 1961.) Consequently, you need to be able to recognize and use both.ĭefinition #3: One atom of the carbon-12 isotope is defined as weighing exactly 12 amu. While the unit u was intended to replace amu, both continue to be used. The ChemTeam will use both u and amu in problem solutions. The most complete definition is a bit more technical (1/12 of the mass of an unbound neutral atom of carbon-12 in its nuclear and electronic ground state and at rest), but the above definition is sufficient for the introductory level.Īn older symbol for atomic mass unit is amu, while the most recent symbol is u (a lower case letter u) which comes from the term 'unified atomic mass unit.' Unified comes from a 1961 agreement between the chemistry and physics communities to use the same definition for the atomic mass unit. Keeping the units in the calculations is annoying, I know, but it helps guide the operations and if you wind up with the unit desired, there is a good chance you've done the problem correctly.There are a number of terms you need to know, at the same time, in order to know what the term "molecular weight" means.ĭefinition #1: The molecular weight of a substance is the weight in atomic mass units of all the atoms in a given formula.ĭefinition #2: An atomic mass unit is defined as 1/12 the weight of one atom of the carbon-12 isotope. That's slightly above Avogadro's number, which is what we expected. Multiply 1.39 moles times the definition of a mole to arrive at the actual number of water molecules:ġ.39 (moles water) * 6.02 x 10 23 molecules water/(mole water) = 8.36 x 10 23 molecules water. The grams cancel and moles moves to the top, leaving moles of water. To find the exact number, divide the available mass (25.0g) by the molar mass (18.0g/mole). We have 25.0 grams of water, so we have more than one mole of water molecules. Think about that before we answer the question. It takes 18 grams of H 2O to obtain 1 mole, or 6.02 x 10 23 molecules of water. We need to molar mass of H 2O, which we find by adding the atomic masses of all the elements, including the number of times they appear. To answer the question of how many molecules of H2O there are in 25.00 grams of water, pay close attention to the units. If you have 1.0 grams of H atoms, you have one mole of H atoms. In both cases, these may be written as 16.0 g/mole and 1.0 grams/mole. Oxygen's atomic weight is 16.0 amu and hydrogen is 1.0 amu. Each 12.0 grams of carbon contain one mole, or 6.02 x 10 23, of carbon atoms. It is an amazingly useful fact that the atomic mass of each element is also what is called it's "molar mass. It is one of the most useful tools in science because it allows conversion of a compound or element's mass into a count of how many molecules/atoms are present in that sample.įor example, carbon, C, has an atomic mass of 12.0 amu (atomic mass units), a number that can be found on all periodic tables. 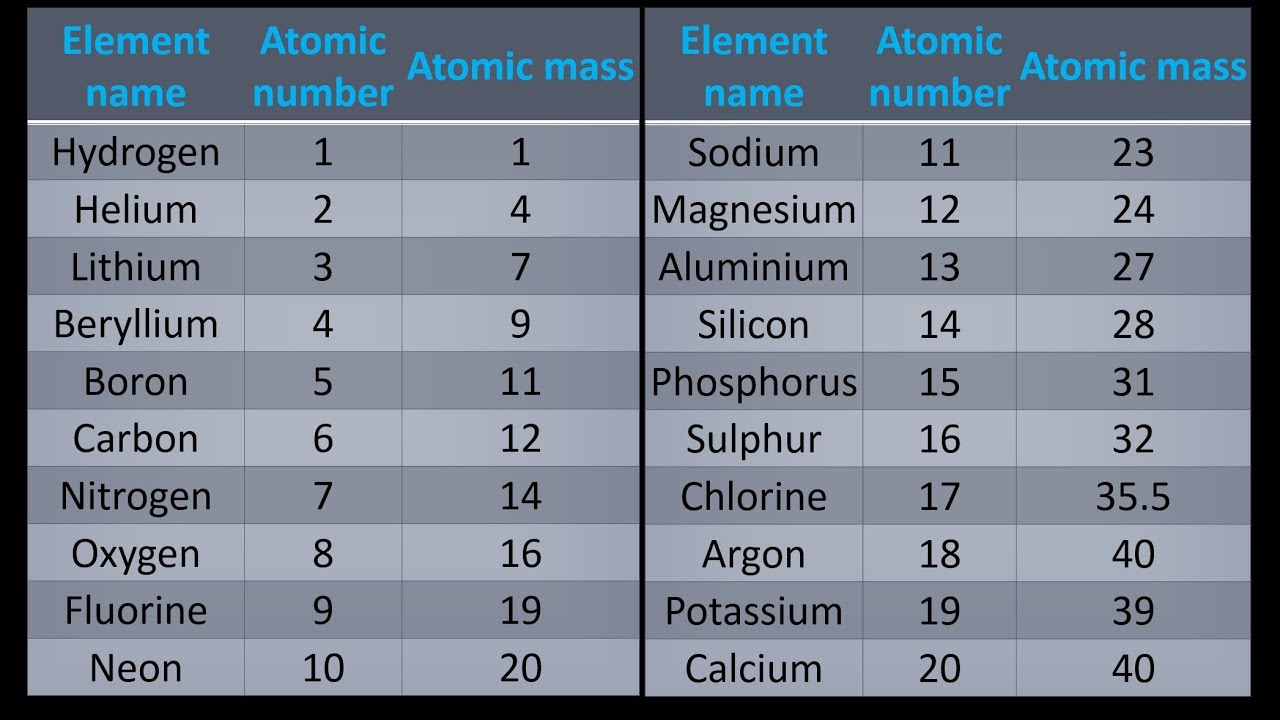
A mole is a nickname for Avogadro's number, 6.02 x 10 23. This question deals with the concept of the mole.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
